Updated: February 4, 2026
Urinary Catheters
Urinary catheters are hollow tubes inserted through the urethra into the bladder to drain urine. The tubing is firm enough to advance but flexible enough to follow the urethral pathway. Catheters help people who are unable to empty their bladder on their own. When the bladder does not empty regularly, urine can build up, cause discomfort, and increase the risk of complications, including kidney problems.
Catheter use is often temporary, such as after surgery or an acute illness. Some injuries or chronic urologic conditions, however, may require long-term or even permanent catheterization. Catheters are available in many sizes, materials, and designs so clinicians can match the device to each patient’s needs.
The following sections review how modern urinary catheters are constructed, how they are used, and how different designs compare, including features found in Cure catheters. A side-by-side comparison chart highlights several widely used catheter models.
Indications
Urinary catheters are generally used when normal bladder emptying is not possible or safe.
- Prolonged immobilization
- Loss of voluntary bladder control
- Urinary incontinence
- Acute urinary retention
- Bladder outlet obstruction
- To aid healing of open perineal or sacral wounds
- To help prevent skin breakdown from constant moisture
- Urologic surgical procedures
- Prolonged surgical procedures
- Surgeries with large-volume infusions or diuretics
Contraindications
Certain findings suggest that catheter insertion may be unsafe.
- Suspected urethral trauma
- Scrotal hematoma
- Pelvic fracture with possible urethral injury
- High-riding prostate on exam
- High suspicion of a urethral tear
- Blood at the urethral meatus
Inappropriate Uses
Catheters should not be used when safer alternatives are available.
- Substitute for routine nursing care in individuals with incontinence
- Obtaining urine samples when the person can urinate voluntarily
Catheter Construction
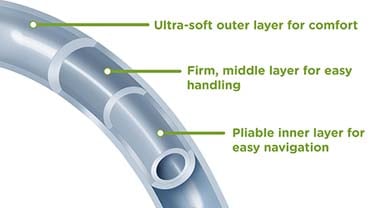
Urinary catheters are most often made from three materials: polyvinyl chloride (PVC), silicone, and latex. Silicone and latex are the most common materials used for indwelling urinary catheters. Many models also have a surface coating to make insertion smoother and to help lower infection risk. Common coatings include hydrophilic layers, silicone-elastomer, polytetrafluoroethylene (PTFE), silver-based finishes, and PTFE-type Teflon. These coatings reduce friction during use and may help decrease irritation and inflammation.
PVC
Polyvinyl chloride (PVC) is a medical-grade plastic used in many healthcare products, including tubing, exam gloves, urine leg bags, and blood collection bags. In catheter form, PVC is clear, which allows staff to see urine color and flow. It is soft and pliable enough to pass through the urethra while remaining firm enough to guide into the bladder.
Advantages
- Smooth eyelets or drainage holes to reduce resistance during insertion
- Clear material for visual observation
- Latex-free construction
- Color-coded funnels to identify French size
- Funnels that support gripping and drainage control
Disadvantages
- May contain plasticizers such as DEHP or DiNP, which are under review for potential health risks
- Manufacturing and disposal can impact the environment
- Concerns exist about long-term exposure to some plasticizers in sensitive populations
Top-Selling PVC Brands
Silicone
Silicone is an inert synthetic polymer used widely in long-term medical devices. It is flexible, stable in a range of temperatures, and resistant to many chemicals. Silicone catheters are biocompatible, meaning the material is designed to limit interaction with body fluids and tissues. This helps support comfort and durability during use.
Advantages
- Firm yet flexible shaft for controlled insertion
- Resists reaction with body fluids
- Biocompatible material
- Less likely to irritate cells and tissues
- Less prone to mineral deposits on the surface
- Surface that can be less favorable to bacterial adherence
- Latex-free construction
- Often selected for longer indwelling use when clinically appropriate
Disadvantages
- Softer structure can be more prone to kinking or deformation in some situations
- Rare reports of catheter tip damage or breakage
- May not be suitable for all patients, depending on individual sensitivity
- Typically higher cost than latex options
Top-Selling Silicone Brands
Latex
Latex catheters are soft and highly flexible, which can support comfort and ease of placement. They are often a lower-cost option. Many latex catheters include an outer coating, such as silicone, silicone elastomer, or hydrogel, to help reduce friction and limit surface irritation.
Advantages
- Very flexible material
- Generally easy to insert when properly lubricated
- Often less expensive than other materials
- Commonly used for short- to moderate-duration catheterization when appropriate
Disadvantages
- Surface can be prone to mineral deposits or encrustation over time
- Encrustation may increase friction and discomfort during removal
- Latex allergy or sensitivity is common in some patient groups
- Higher risk of hypersensitivity reactions in individuals with latex sensitivity
- May be more irritating to the urinary tract than latex-free options in some cases
Top-Selling Latex Catheters

Catheter Coatings
Coatings
Urinary catheters are commonly used in chronic care facilities and senior facilities throughout the world. Many of the problems associated with catheters include infection, encrustation, tissue trauma, and inflammation. These issues can be correlated to the catheter surface. Many of the shortcomings with catheter surfaces can be alleviated by simply adding a friction-reducing coating. Several coatings are now in use, including hydrogel, Teflon,® and silicone. Hydrophilic coatings appear to be the best option. Hydrogel provides a smooth, soft surface with lubricating properties.21
Hydrophilic
A hydrophilic surface coating is a polymer layer that readily absorbs liquid. When immersed in water, it swells to a smooth, slick film. The film provides for easier insertion into the body. A hydrogel-coated latex urinary catheter has a smooth surface with fissures, ripples, and furrows filled with a slick cover. The topography of the hydrophilic coated latex surface compares with a 100% silicone catheter.22
Silicone
A silicone coating provides a smooth surface. A silicone outer layer on a latex catheter provides a smooth covering to help reduce patient discomfort and irritation during insertion.
Silicone-Elastomer
A silicone-elastomer coating is a hydrophobic material that repels moisture. This layer helps to protect the urethral from irritation. It offers a glassy surface for a less painful insertion.
Polytetrafluoroethylene
Polytetrafluoroethylene (PTFE) or Teflon is a fluorocarbon-based polymer that lines the catheter to reduce friction by providing a smooth surface. PTFE coatings increase torque control, flexion, and stiffness to provides easier insertion.
Antimicrobial Agents
Silver is an antibacterial agent. Adding silver or another antimicrobial such as chlorhexidine, Triclosan, enzymes, nitric oxide, or liposomes to the coating reduces the risk of infection and inflammation. Antimicrobial coatings help protect from urinary tract infections (UTI) that often plaque long-term catheterization patients.
More than 75% of hospital-acquired or nosocomial urinary tract infections began by urinary catheters. These devices treated 15 to 25% of hospitalized patients. Among other purposes, urinary catheters are primarily used for draining urine after surgeries and for urinary incontinence. During catheter-associated urinary tract infections, bacteria travel up to the bladder and cause infection.23

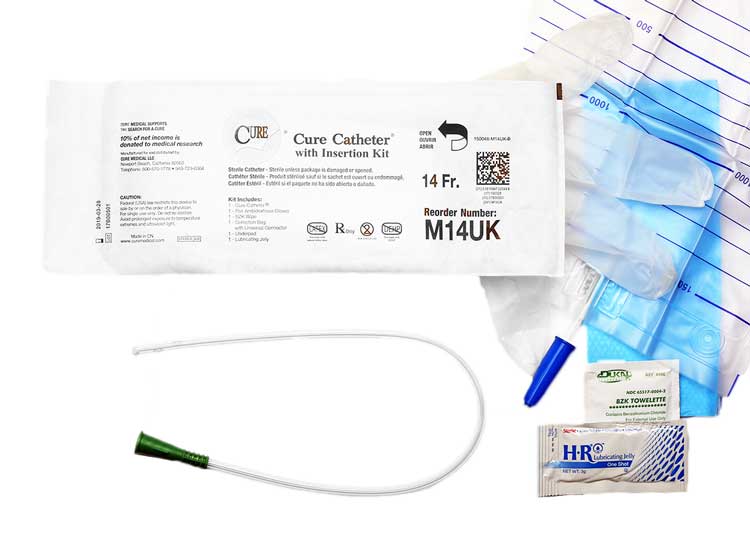
The Cure Medical Catheter Difference
Having discussed what catheterization is, the indications for use, contraindications, construction, phthalates, and the various coating options, we will now turn to the role of Cure catheters and how they compare with the other brand offerings. Cure catheters offer significant advantages over other branded products. These advantages include PVC construction but without the use of DEHP, DINP, BPA, or natural rubber latex. Cure catheters do not kink when bent. They come with smooth, polished eyelets that make them easy to insert and extract. Packaged ready-to-use, the packing material is minimal for a lower environmental impact. Below is a summary of the Cure benefits and features.
Advantages of Cure Catheters
- ✓ Smooth, polished eyelets
- ✓ Packaged ready-to-use
- ✓ Straight or coude options
- ✓ Male and female options
- ✓ Sizes 8 to 18 French
- ✓ Pre-lubricated (Ultra, Hydrophilic) options
- ✓ CoverAll application
- ✓ No roll connector funnel facilitates draining
- ✓ Will not kink when bent
- ✓ DEHP-free
- ✓ DINP-free
- ✓ BPA-free
- ✓ Natural Rubber Latex-free
- ✓ Small, flexible packaging
- ✓ Minimal disposal material
Comparing the Top Catheter Brands
| Intermittent | Ultra | Closed System |
SpeediCath Compact |
VarPro Plus |
Apogee | GentleCath | Magic3 | Red Rubber Robinson |
|
| Manufacturer | Cure | Cure | Cure | Coloplast | Holister | Holister | ConvaTec | Bard | Teleflex |
| Construction | PVC | PVC | PVC | PVC | PVC | PVC | PVC | Silicone | Latex |
| Coating Options | Hydrophilic | Hydrophilic | Hydrophilic | Hydrophilic | Hydrophilic | Hydrophilic | Hydrophilic | Hydrophilic Antibacterial |
- |
| Eyelets | Smooth & Polished |
Smooth & Polished |
Smooth & Polished |
Smooth & Fire Polished |
Smooth | Smooth & Fire Polished |
Polished | 4-small | 2 |
| Tip Options | Straight Coude |
Straight Coude |
Straight Coude |
Straight Coude |
Straight | Straight Coude |
Straight Tiemann |
Straight | Straight |
| No-Drip | Yes | Yes | Yes | - | - | - | - | - | - |
| CoverAll | Yes | Yes | Yes | - | - | - | - | - | - |
| Easy Gripper Sleeve | Yes | Yes | Yes | - | - | - | - | - | - |
| No Roll Funnel | Yes | Yes | Yes | - | - | - | - | - | - |
| Kink-Free | Yes | Yes | Yes | - | - | - | - | Resistant | - |
| DEHP-Free | Yes | Yes | Yes | Yes | Yes | - | Yes | Yes | - |
| DINP-Free | Yes | Yes | Yes | Yes | Yes | - | - | Yes | - |
| BPA-Free | Yes | Yes | Yes | Yes | Yes | - | - | Yes | - |
| NR Latex-Free | Yes | Yes | Yes | - | Yes | Yes | Yes | Yes | No |
| Other Features | BZK Wipe Swabsticks Gloves Sterile Wipe Underpad |
Pull-Ring | Anti-Reflux Valve |
Introducer Tip |
No-Touch Sleeve |
3-layers | X-ray Opaque |
||
| Introducer Tip |
Adhesive Dot |
Hydration | FeelClean Technology |
||||||
| Integrated Bag |
Integrated Bag |
Integrated Bag |
Product Videos
Dr. Gerard Henry on DEHP Video (1:21 minutes)
Hydrophilic Cure Video (1:29 minutes)
Cure Ultra Video (1:11 minutes)
Cure Ultra Coude Video (1:11 minutes)
Footnotes
- 1 Guideline for Prevention of Catheter-Associated Urinary Tract Infections. Infection Control. Centers for Disease Control and Prevention. (2009) Page 2. (Last Accessed September-9-2020)
- 2 Urinary Catheter Insertion. Urinary Catheter. Department of Emergency Medicine. University of Ottawa. (Last Accessed September-9-2020)
- 3 Guidelines, Page 2. (Last Accessed September-9-2020)
- 4 Moneret-Vautrin, Denise-Anne, et al. Prospective study of risk factors in natural rubber latex hypersensitivity. Journal of allergy and clinical immunology 92.5 (1993): Page 668. (Last Accessed September-9-2020)
- 5 Deval, Ravi, et al. Natural rubber latex allergy. Indian Journal of Dermatology, Venereology, and Leprology 74.4 (2008): 304. (Last Accessed September-9-2020)
- 6 Tamura, N. K., A. Gasparetto, and T. I. E. Svidzinski. Evaluation of the adherence of Candida species to urinary catheters. Mycopathologia 156.4 (2003): 269-272. (Last Accessed September-9-2020)
- 7 Woodward, Sue. Complications of allergies to latex urinary catheters. British Journal of Nursing 6.14 (1997): 786-793. (Last Accessed September-9-2020)
- 8 Lawrence, E. L., and I. G. Turner. Materials for urinary catheters: a review of their history and development in the UK. Medical engineering & physics 27.6 (2005): 443-453. (Last Accessed September-9-2020)
- 9 Marie, Cécile, et al. Exposure of hospitalised pregnant women to plasticizers contained in medical devices. BMC women's health 17.1 (2017): 45. (Last Accessed September-9-2020)
- 10 Koch, Holger M., Ralf Preuss, and J. Di Angerer. Di (2‐ethylhexyl) phthalate (DEHP): human metabolism and internal exposure–an update and latest results 1." International journal of andrology 29.1 (2006): 155-165. (Last Accessed September-9-2020)
- 11 Tickner, Joel A., et al. Health risks posed by use of Di‐2‐ethylhexyl phthalate (DEHP) in PVC medical devices: A critical review. American journal of industrial medicine 39.1 (2001): 100-111. (Last Accessed September-9-2020)
- 12 ToxFAQs for Di(2-ethylhexyl)phthalate (DEHP). Toxic Substances Portal. Centers for Disease Control. (Last Accessed September-9-2020)
- 13 Diisononyl Phthalate (DiNP). Vermont Department of Health. (Last Accessed September-9-2020)
- 14 Wang, Yu, Hongkai Zhu, and Kurunthachalam Kannan. A review of biomonitoring of phthalate exposures. Toxics 7.2 (2019): 21. (Last Accessed September-9-2020)
- 15 CONSUMER PRODUCT SAFETY IMPROVEMENT ACT OF 2008. PUBLIC LAW 110–314—AUG. 14, 2008. Consumer Product Safety Improvement Act of 2008. Commerce and trade. 15 USC 2051. (Last Accessed September-9-2020)
- 16 vom Saal, Frederick S., and Wade V. Welshons. Evidence that bisphenol A (BPA) can be accurately measured without contamination in human serum and urine, and that BPA causes numerous hazards from multiple routes of exposure. Molecular and cellular endocrinology 398.1-2 (2014): 101-113. (Last Accessed September-9-2020)
- 17 Konieczna, Aleksandra, Aleksandra Rutkowska, and D. Rachon. Health risk of exposure to Bisphenol A (BPA). Roczniki Państwowego Zakładu Higieny 66.1 (2015). (Last Accessed September-9-2020)
- 18 Bisphenol A (BPA). National Institute of Environmental Health Sciences. (Last Accessed September-9-2020)
- 19 Bisphenol A (BPA) Factsheet. National Biomonitoring Program. Centers for Disease Control. (Last Accessed September-9-2020)
- 20 Cho, Seung Hee, et al. Urinary bisphenol A versus serum bisphenol A concentration and ovarian reproductive outcomes among IVF patients: Which is a better biomarker of BPA exposure?. Molecular & Cellular Toxicology 13.4 (2017): 351-359. (Last Accessed September-9-2020)
- 21 Graiver, D., R. L. Durall, and T. Okada. Surface morphology and friction coefficient of various types of Foley catheter. Biomaterials 14.6 (1993): 465-469. (Last Accessed September-9-2020)
- 22 Cox, A. J. Effect of a hydrogel coating on the surface topography of latex-based urinary catheters: an SEM study. Biomaterials 8.6 (1987): 500-502. (Last Accessed September-9-2020)
- 23 Singha, Priyadarshini, Jason Locklin, and Hitesh Handa. A review of the recent advances in antimicrobial coatings for urinary catheters. Acta biomaterialia 50 (2017): 20-40. (Last Accessed September-9-2020)
Cure Medical Literature
 Instruction Guide for catheterization for women.
Instruction Guide for catheterization for women. Instructions for women using closed system.
Instructions for women using closed system. Instructions for Men for intermittent catheterization.
Instructions for Men for intermittent catheterization. Hydrophilic Caterization instructions for men.
Hydrophilic Caterization instructions for men. Closed System Insertion instructions for men.
Closed System Insertion instructions for men. Intermittent Closed System Catheters instructions for parents of girls.
Intermittent Closed System Catheters instructions for parents of girls. Use Instructions for Intermittent Closed System Catheters for parents of boys.
Use Instructions for Intermittent Closed System Catheters for parents of boys. Free Cure Catheter Samples see and feel the difference.
Free Cure Catheter Samples see and feel the difference.
Medical Studies
 Silva, Manori J., et al. Urinary oxidative metabolites of di (2-ethylhexyl) phthalate in humans. Toxicology 219.1-3 (2006): 22-32. (Last Accessed September-9-2020)
Silva, Manori J., et al. Urinary oxidative metabolites of di (2-ethylhexyl) phthalate in humans. Toxicology 219.1-3 (2006): 22-32. (Last Accessed September-9-2020) Vesterberg, Anna, Magnus Hedenmark, and A. M. Vass. PVC in medical devices. An Inventory of PVC and Phthalates Containing Devices Used in Health Care (2005). (Last Accessed September-9-2020)
Vesterberg, Anna, Magnus Hedenmark, and A. M. Vass. PVC in medical devices. An Inventory of PVC and Phthalates Containing Devices Used in Health Care (2005). (Last Accessed September-9-2020) Johns, Lauren E., et al. Exposure assessment issues in epidemiology studies of phthalates. Environment international 85 (2015): 27-39. (Last Accessed September-9-2020)
Johns, Lauren E., et al. Exposure assessment issues in epidemiology studies of phthalates. Environment international 85 (2015): 27-39. (Last Accessed September-9-2020) California Proposition 65. State of California Legistature. (1987) (Last Accessed September-9-2020)
California Proposition 65. State of California Legistature. (1987) (Last Accessed September-9-2020) Phthalates Factsheet. National Biomonitoring Program. Centers for Disease Control. (Last Accessed September-9-2020)
Phthalates Factsheet. National Biomonitoring Program. Centers for Disease Control. (Last Accessed September-9-2020) Ikarashi, Yoshiaki, et al. Comparative studies by cell culture and in vivo implantation test on the toxicity of natural rubber latex materials. Journal of biomedical materials research 26.3 (1992): 339-356 (Last Accessed September-9-2020)
Ikarashi, Yoshiaki, et al. Comparative studies by cell culture and in vivo implantation test on the toxicity of natural rubber latex materials. Journal of biomedical materials research 26.3 (1992): 339-356 (Last Accessed September-9-2020) Graham, D. T., et al. In vivo validation of a cell culture test for biocompatibility testing of urinary catheters. Journal of biomedical materials research 18.9 (1984): 1125-1135. (Last Accessed September-9-2020)
Graham, D. T., et al. In vivo validation of a cell culture test for biocompatibility testing of urinary catheters. Journal of biomedical materials research 18.9 (1984): 1125-1135. (Last Accessed September-9-2020) Ha, U-Syn, and Yong-Hyun Cho. Catheter-associated urinary tract infections: new aspects of novel urinary catheters. International journal of antimicrobial agents 28.6 (2006): 485-490. (Last Accessed September-9-2020)
Ha, U-Syn, and Yong-Hyun Cho. Catheter-associated urinary tract infections: new aspects of novel urinary catheters. International journal of antimicrobial agents 28.6 (2006): 485-490. (Last Accessed September-9-2020) Johansson, Kerstin, et al. Evaluation of a new PVC-free catheter material for intermittent catheterization: a prospective, randomized, crossover study. Scandinavian journal of urology 47.1 (2013): 33-37. (Last Accessed September-9-2020)
Johansson, Kerstin, et al. Evaluation of a new PVC-free catheter material for intermittent catheterization: a prospective, randomized, crossover study. Scandinavian journal of urology 47.1 (2013): 33-37. (Last Accessed September-9-2020) Koch, Holger M., et al. Internal exposure of the general population to DEHP and other phthalates—determination of secondary and primary phthalate monoester metabolites in urine. Environmental research 93.2 (2003): 177-185. (Last Accessed September-9-2020)
Koch, Holger M., et al. Internal exposure of the general population to DEHP and other phthalates—determination of secondary and primary phthalate monoester metabolites in urine. Environmental research 93.2 (2003): 177-185. (Last Accessed September-9-2020) Koch, Holger M., et al. Di (2-ethylhexyl) phthalate (DEHP) exposure of voluntary plasma and platelet donors. International journal of hygiene and environmental health 208.6 (2005): 489-498. (Last Accessed September-9-2020)
Koch, Holger M., et al. Di (2-ethylhexyl) phthalate (DEHP) exposure of voluntary plasma and platelet donors. International journal of hygiene and environmental health 208.6 (2005): 489-498. (Last Accessed September-9-2020) Stojanoska, Milica Medic, et al. Do diethyl phthalate (DEP) and di-2-ethylhexyl phthalate (DEHP) influence the metabolic syndrome parameters? Pilot study. Environmental monitoring and assessment 187.8 (2015): 526. (Last Accessed September-9-2020)
Stojanoska, Milica Medic, et al. Do diethyl phthalate (DEP) and di-2-ethylhexyl phthalate (DEHP) influence the metabolic syndrome parameters? Pilot study. Environmental monitoring and assessment 187.8 (2015): 526. (Last Accessed September-9-2020) Gray Jr, L. Earl, et al. Perinatal exposure to the phthalates DEHP, BBP, and DINP, but not DEP, DMP, or DOTP, alters sexual differentiation of the male rat. Toxicological Sciences 58.2 (2000): 350-365. (Last Accessed September-9-2020)
Gray Jr, L. Earl, et al. Perinatal exposure to the phthalates DEHP, BBP, and DINP, but not DEP, DMP, or DOTP, alters sexual differentiation of the male rat. Toxicological Sciences 58.2 (2000): 350-365. (Last Accessed September-9-2020) Stripple, Håkan, Robert Westman, and Daniel Holm. Development and environmental improvements of plastics for hydrophilic catheters in medical care: an environmental evaluation. Journal of Cleaner Production 16.16 (2008): 1764-1776. (Last Accessed September-9-2020)
Stripple, Håkan, Robert Westman, and Daniel Holm. Development and environmental improvements of plastics for hydrophilic catheters in medical care: an environmental evaluation. Journal of Cleaner Production 16.16 (2008): 1764-1776. (Last Accessed September-9-2020) Phthalates Factsheet. National Biomonitoring Program. Centers for Disease Control. (Last Accessed September-9-2020)
Phthalates Factsheet. National Biomonitoring Program. Centers for Disease Control. (Last Accessed September-9-2020)

Login and Registration Form