

Intranasal Transmucosal Drug Delivery











Intranasal medication is a procedure for administering medications through nasal passages. Also known as transmucosal administration or nasal drug delivery, intranasal medicating employs an atomization device that fragments the medication into fine particles as it is being sprayed into the nose. This method of administration offers several advantages. This article will examine these advantages as well as what medications are commonly dispensed using this method.
Advantages of Transmucosal Administration
The use of nasal administrated medications offers 6 key advantages. Nasal administration provides a direct route for medications, avoiding gastrointestinal destruction and hepatic first-pass metabolism. Transmucosal medicating effectively increases bioavailability while decreasing the time of onset. The process is easy to use and safe. We will explore these six advantages in more detail below.
Direct Route
Nasal administration is a nose-brain pathway for much more rapid dispersal. The olfactory mucosa is in direct contact with the brain and cerebrospinal fluid (CSF). When medications are absorbed through the olfactory mucosa, they are directly transported to the CSF. This nose-to-brain pathway offers a rapid and direct route for medication delivery to the brain.
Gastrointestinal Destruction Prevention
Drugs administered orally are absorbed through the gastrointestinal tract. While enroute, the drug can be significantly degraded by gastric acidity, intestinal membrane enzymes, complexion with food constituents, or bacterial enzymes. Oral medications sit in the stomach for 30 to 45 minutes before being sent to the intestines for absorption. After absorption into the blood, the medication is delivered to the liver for detoxification. This time-consuming digestive process destroys much of the medication before it reaches the bloodstream for dispersal throughout the body. While oral administration remains the most convenient delivery method, it provides very little absorption and transfer of the drug to the bloodstream.
Hepatic First Pass Metabolism
Nasal medication bypasses first-pass metabolism while oral medications do not. As mentioned above, all blood from the intestines is routed to the liver for detoxification. The liver metabolizes around 90% of oral medications before the medication reaches the heart. This leaves very little of the drug remaining after liver detoxification for patient use. A nasal administered drug; however, absorbs directly into the veins through the nasal mucosa; therefore, by-passing first-pass metabolism. The heart then pumps the drug to the entire body without delay.
Bioavailablity
Bioavailability refers to the number of drugs transmitted into the bloodstream. For instance, IV-administered medications have 100% bioavailability while most oral medications have only around 5 to 10% bioavailability. The bioavailability of nasal medications is dependent upon the molecule size and pH of the medication. For instance, Nalozon, a popular intranasal medication, has 90+% bioavailability while midazolam has 75+% Nalozone and Fentanyl has 80+%. Transmucosal administration is generally more effective than subcutaneous or intramuscular injections.
Time of Onset
Intranasal administration is rapidly effective with an onset time between only 2 to 10 minutes.
Ease of Use
Nasally administered drugs are convenient and easy. The administration is painless and does not expose the patient or caregiver to accidental needle sticks. Waste is reduced without the need for sharps containers and hazardous disposal. Intranasal administration is safe for the patient with no high peak serum levels. Nasally administered drugs are safer to use for violent individuals or for seizure victims. Various medications may be delivered to the patient in any position, whether sitting, lying down, prone, on the side, or upside-down. Since nasal administration takes only a second to administer the patient may not need to be restrained.
Types of Nasal Mucosa Administrations
- Spray Bottles
- Nose Droppers
- Pressurized Aerosol
- Atomized Unit Dose
Nasal administrations may take the form of spray bottles, nose droppers, pressurized aerosol, or atomized unit dose sprays. The latter is the most effective method. Spray bottles fail to provide a consistent dosage and often result in excessive amounts of the drug draining into the throat. Large particles of the medication can also form deposits on the nasal mucosa. Nose droppers often produce the same unwanted results as spray bottles with much of the medication not being absorbed and draining into the throat. Pressurized aerosol devices tend to break the medication into smaller particles that flow down to the lungs which offer less effectiveness with absorption and may result in respiratory complications.
Atomization of the medication reduces the particles to 2 to 10 micrometers which provides for optimal absorption through the nasal mucosa. The mucosa offers a large surface area of more than 180 square centimeters of highly vascularized tissue. This area has more blood vessels per gram of tissue than muscle tissue, the brain, or even the liver. Absorption through the nasal mucosa offers the best results for rapidity and efficacy. Atomization best reduces the medication particles to the optimal size for absorption. Additionally, because the medication is atomized into a mist form, it less is likely to be expelled from the nose into the external environment.
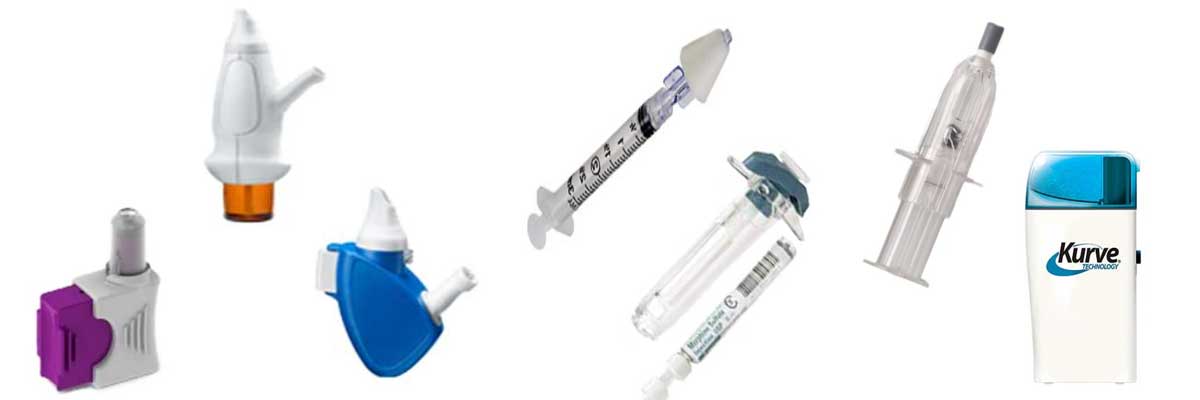
Nasal Medication Delivery Devices
There are several manufacturers that make nasal medication devices. Below is a list of nasal administration devices listed alphabetically.
Top Best Nasal Medication Delivery Devices
- Accuspray by Becton Dickenson - offers patients a less invasive drug administration. Designed for use by both doctors and patients, Accuspray is easy-to-use with no pre-activation or loading required. The drug to be administered is clearly visible, allowing visual inspection of the medication before administration. Accuspray provides for the administration to be easily divided, allowing the drug to be delivered to each nostril to access more mucosa membranes for better dispersal.
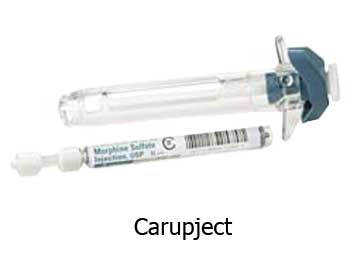
- Carpuject by Pfizer - Carpuject has a compact design that is needleless and prevents injuries. The Carpuject syringe system can dispense a wide variety of medications with easy-loading cartridges. Caregivers enjoy easy one-handed operation and disposal.
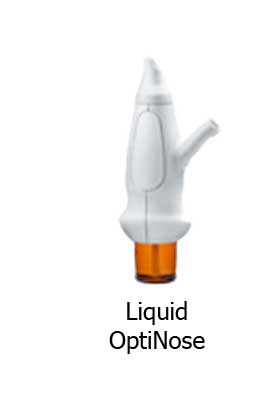
- OptiNose - The OptiNose nasal device is designed to treat chronic nasal inflammatory diseases, migraines, and Autism Spectrum disorder. OptiNose provides two separate delivery devices designed to administer either powder or liquid medications. The devices minimize local irritation and allow patients to self-medicate.
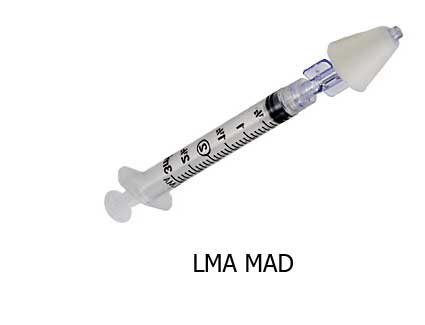
- MAD Intranasal Mucosal Atomization Device by Teleflex – This nasal atomization device is designed to be used with drugs approved for intranasal delivery. It requires no sterile procedures, is painless, and is quick and easy to administer. The atomized medication is rapidly absorbed across the mucosal membranes directly into the bloodstream for greater bioavailability.
- CPD by Kurve Technology - This nasal atomization device is non-invasive and provides controlled particle dispersion to deliver medications directly to the brain. The CPD nasal drug delivery system can control the size of the liquid droplets down to 1 micron regardless of the viscosity of the formulation.
- DirectHaler by Acerus Pharma - The DirectHaler is also known as the Direct-Haler and is a nasal drug delivery device designed to use the patient's breath to disperse dry-powered medications to the patient. Recently, the Acerus Pharmaceutical Corporation acquired this award-winning intranasal drug delivery device from a company in Denmark.
- VeriDoser by Mystic Pharmaceuticals - The VeriDoser offers nasal drug delivery with precise, calibrated dosing. Mystic's VeriDoser requires no priming and has a dose counter. The spray plume is configurable into steam, spray, or aerosolized spray. The design accommodates proteins, peptides, and stem cell formulations to treat CNS, neurodegenerative disorders, brain tumors, HIV encephalopathy, epilepsy, Huntington's, Alzheimer's, and Parkinson's.
Common Medications Dispensed Using Nasal Atomization
Nasal atomization delivery is being developed for many applications, including Alzheimer's, Parkinson's, brain tumors, diabetes, multiple sclerosis, schizophrenia, pain control, and migraines. Below is a list of the most popular drugs currently dispensed by intranasal administration grouped by treatment type.
Pain Control - Opiates
Fentanyl
Sufentanyl
Ketamine
Sedation - ά-2 Agonists
Midazolam
Dexmedetomidine
Seizure Therapy – Benzodiazepines
Larazepam
Midazolam
Opiate Overdose
Naloxone (Narcan)
Motion Sickness
Scopolamine
Reversal of Conscious Sedation
Romazicon
Urination Suppression
DDAVP (Desmopressin)
Angina (Chest Pain)
Nitroglycerin
Migraines
Sumatriptan
Specific Nasal Drug Delivery Administrations
Fentanyl
Fentanyl is an opiate used for pain control and is an ideal drug for use with intranasal administration. A prescription is required for these drugs. Fentanyl has a rapid onset and short duration and is one of the strongest opiate drugs sold on the market. Intranasal fentanyl is rapidly absorbed, reaching a maximum concentration in just 4 to 11 minutes. Bioavailability is around 71% but appears to be pH dependent with increased bioavailability associated with higher pH. 1
Ketamine
Ketamine is a dissociative anesthetic opiate that is often used for treatment-resistant depression. Besides IV administration, intranasal is the second most common method of dispensing Ketamine. Intranasal administration requires less skill to use and offers less risk to the patient.
Midazolam
Midazolam produces sleepiness or drowsiness and helps relieve anxiety. It also produces amnesia so that the patient will forget any discomfort or pain that may occur following surgery. In intensive care hospital situations, Midazolam is used to induce unconsciousness. This drug is also used to treat epilepsy and seizures. Midazolam requires a prescription and close doctor supervision. Intranasal midazolam was found in a study to have the same efficacy as IV administrations but that the short administration time for nasal, as opposed to IV setup, favored the use of nasal delivery. 2
Narcan Usage
Narcan is an FDA-approved drug known as naloxone hydrochloride. This drug is an opioid antagonist used for the emergency treatment of individuals with respiratory or central nervous system failure due to opioid overdose. Administration of naloxone by nasal spray atomization is easy and convenient and offers increased efficacy. Narcan nasal spray is designed to provide a single 4 mg dose. Administration of the drug using a Narcan MAD Syringe allows the medication to be atomized for quick metabolism. 3 The LMA MAD Nasal Intranasal Mucosal Atomization Device from Teleflex is specifically designed for administering this lifesaving drug.
First Responders Use of Intranasal Naloxone
Emergency Medical Technicians (EMTs) and ambulance crews have found the use of Narcan nasal spray to restore spontaneous respiration for drug overdose patients in just a matter of seconds. Ease of use without the risk of needlestick injury is cited as a primary benefit. The requirement for minimal training has also been cited as a benefit. Some have advocated extending the use of Narcan nasal sprays to community workers that often come into contact with drug abusers.4 Many police departments are now equipping officers with opioid-overdose antidotes because the police are often the first on the scene.5 U.S. Attorney General Eric Holder advocates that the police and fire departments should equip their first responders with naloxone to save drug overdose victims.6
Parental Use of Nasal Nacan
Some states are allowing Naloxone to be sold at drug stores without a prescription in an effort to curb teenage overdose deaths. 7
Standard Drug Administration Devices For Sale
Summary
Intranasal medication is a procedure for administering medications nasally. This method of administration offers a direct route to the olfactory mucosa, prevents gastrointestional destruction, offers bioavailability, ease of use, and other advantages. Widely used by EMT's and law enforcement, nasal administration is growing as a life saving measure and as a method for fast drug delivery.
Footnotes
1Foster, David, et al. Pharmacokinetics and pharmacodynamics of intranasal versus intravenous fentanyl in patients with pain after oral surgery. Annals of Pharmacotherapy 42.10 (2008): 1380-1387.
2Narcan.com
4Strang, John, et al. Clinical provision of improvised nasal naloxone without experimental testing and without regulatory approval: imaginative shortcut or dangerous bypass of essential safety procedures? Addiction (2016).
5Fuller, Steve. City police make first Narcan save. The Ellsworth American. August 24, 2016.
6Sledge, Matt. Eric Holder Calls On First Responders To Carry Naloxone, Anti-Overdose Drug. The Huffington Post. April 16, 2014.
7Fiangan, Katlin. Narcan to Be Sold at Pharmacies Without Prescription in Vermont. NECN.com. August 25, 2016.
Medical Studies
Barton, Erik D., et al. Efficacy of intranasal naloxone as a needleless alternative for the treatment of opioid overdose in the prehospital setting. The Journal of emergency medicine 29.3 (2005): 265-271.
Kelly, A., and Z. Koutsogiannis. Intranasal naloxone for pfe threatening opioid toxicity. Emergency medicine journal: EMJ 19.4 (2002): 375.
Barton, Erik D., et al. Efficacy of intranasal naloxone as a needleless alternative for the treatment of opioid overdose in the prehospital setting. The Journal of emergency medicine 29.3 (2005): 265-271.
Hussain, Anwar A. Intranasal drug delivery. Advanced drug delivery reviews 29.1 (1998): 39-49.
Reger, M. A., et al. Effects of intranasal insulin on cognition in memory-impaired older adults: modulation by APOE genotype. Neurobiology of aging27.3 (2006): 451-458.
Wermepng, Daniel P., and Jodi L. Miller. Intranasal drug delivery. DRUGS AND THE PHARMACEUTICAL SCIENCES 126 (2003): 727-748.

Login and Registration Form