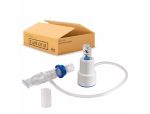
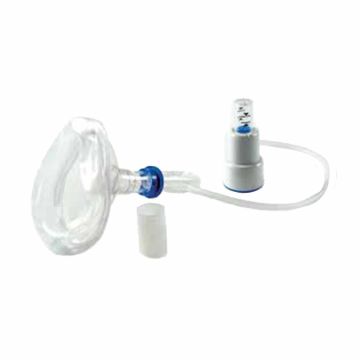
The TheraPEP PEP Therapy System by Smith's Medical is a lightweight, easy-to-use positive expiratory pressure system designed for clearing airways, helping to promote airflow in patients with chronic pulmonary disease (COPD) or cystic fibrosis (CF), as well as individuals recovering from surgery. With the TheraPEP System patients breathe out with a moderate force through resistance which in turn creates a positive pressure that gets beneath obstructive mucus, mobilizing it to the front of the airways so that it can be coughed out.
With the TheraPEP PEP Therapy System, respiratory therapy can be self-administered by the patient in the comfort of their own home, or anywhere or at any time that the need arises, in much less time than the labor-intensive, time-consuming chest physiotherapy (CPT). The device features a built-in pressure indicator, six fixed orifice options, and can be used with a mask or mouthpiece, as well as a nebulizer. The TheraPEP device is sturdy enough to resist breakage, allowing it to go with the user wherever they go.
Vitality Medical also carries other PEP therapy devices, such as the Acapella Duet Nebulizer & Vibratory PEP Therapy Unit and the Acapella Choice Vibratory PEP Mucus Clearance Device.
Product Features and Benefits:
- Accommodates Virtually Any Patient's Lung Capacity.
- Resists Breakage, Unlike Fragile, Costly Manometers.
- Provides Immediate, Visual 360 Degree Feedback of Prescribed Pressure.
- May Be Used With a Mask or Mouthpiece or Nebulizer.
- Allows Inhalation and Exhalation Without Removing From Mask.
- Six Fixed Orifice Options.
- Built-In Durable Pressure Indicator.
- Inspiratory Valve.
Product Specifications:
- Manufacturer: Smiths Medical ASD
- HCPCS Code: E1399
- Product Numbers: 20-1112, 20-3112, 20-5112, 20-7112
Manuals and Documents
 Product Manual contains product care and instructions.
Product Manual contains product care and instructions.
Product Videos
TheraPEP PEP Therapy System Video (2:08 minutes)





Login and Registration Form