3M 1860 N95 Surgical Respirator Mask
Product Highlights
- High bacterial filtration efficiency
- NIOSH, CDC, and FDA-approved
- Not made with natural rubber latex
- Adjustable metal nosepiece
- Braided elastic headbands
- Fluid-resistant
- Fast Delivery
- Easy Returns
- AutoShip eligible
free shipping on $49+
| Item# | Description | Size | Price |
|---|---|---|---|
| 1860 | On Sale!Regular | Box of 20 |
Reg Price: $32.29
Sale Price:
$32.29
($1.61 ea)
You Save: $0.00 (0%)
|
| 1860 | Regular | Case of 120 |
List Price: $143.21
Price:
$131.75
($1.10 ea)
You Save: $11.46 (8%)
|
| 1860S |
Small
Fast 1-3 Day Shipping! |
Box of 20 |
List Price: $29.02
Price:
$26.70
($1.34 ea)
You Save: $2.32 (8%)
|
| 1860S | On Sale!Small | Case of 120 |
Reg Price: $185.98
Sale Price:
$185.98
($1.55 ea)
You Save: $0.00 (0%)
|
Description
3M 1860 N95 Mask Series Overview
Who Uses the 3m 1860 N95 Mask? Surgeons as well as individuals needing N95 mask protection.
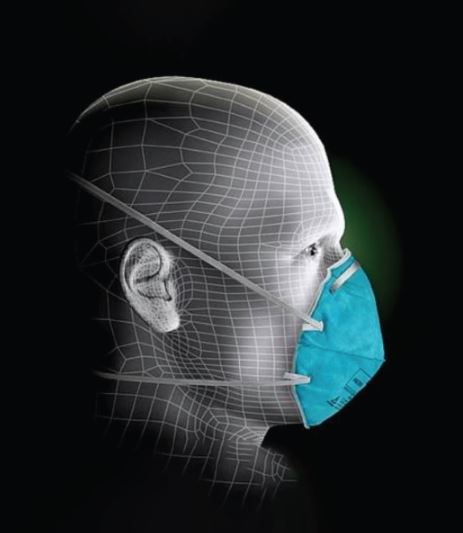
Why They Love It: As an N95, this mask is approved by the FDA for surgical use. It is fluid-resistant and has a high bacterial filtration efficiency (BFE). A bendable metal nose ridge works with two elastic headbands to keep the mask comfortable and snug against the wearer’s face.
The 3M 1860S N95 Mask, and 3M 1860 Mask, have a 99% bacterial filtration efficiency (BFE) and are fluid-resistant. This N95 mask by 3M meets and exceeds the specification set by the National Institute for Occupational Safety and Health (NIOSH) and is therefore safe for use in healthcare environments. In addition to this stamp of approval, the Centers for Disease Control (CDC), certified its effectivenessness in protecting against exposure to Mycobacterium tuberculosis.
Some of the comfort features include a cup-shaped bevel that makes this respirator collapse-resistant. Two braided elastic bands secure the mask to the wearer’s face, and a metal piece bends to the unique shape of the user’s nose ridge. This ensures the mask stays in place for a sure seal, helping to reduce the risk of mask adjustments and touching the face. The mask is not made with natural rubber latex to cause possible facial irritation.
Vitality Medical sells several styles of respirators and other hospital supplies. Scroll to the bottom to compare the 1860 against other N95 masks. Stay comfortably protected with a 3M Health Care Particulate Respirator and Surgical Mask today!
Additional Resources
Fast Shipping & Handling Most orders are processed and shipped within 24 hours!
Key Features

Elastic Head Straps
Secure the mask comfortably in place.


Cup Shape Design
Keeps the mask from collapsing.


Fluid Resistance
Reduces the potential exposure to blood and other body fluids.


Adjustable Nose Clip
Provides a customized fit and helps provide a secure seal.
Product Details
Certification and Compliance
The 1860 N95 Respirator Mask provides confidence with the following certifications and compliance:
- Meets and exceeds the National Institute for Occupational Safety and Health (NIOSH) regulations for its ability to filter 99% of solid and liquid aerosols not containing oils.
- Meets CDC standards and protects against Mycobacterium tuberculosis.
- FDA-approval for use in operating rooms.
Application

Removal
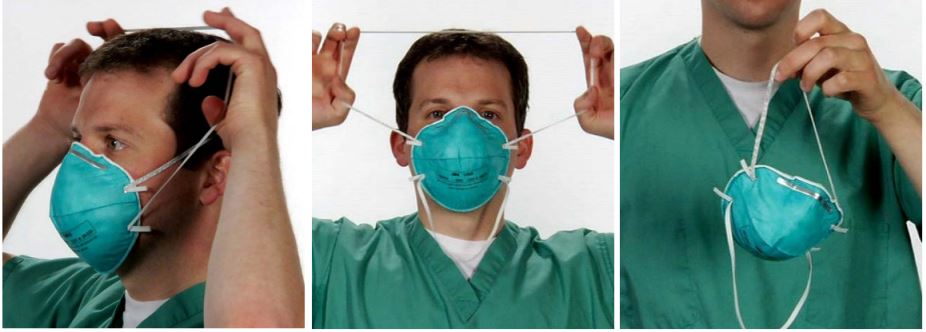

Additional Features
- Meets NIOSH N95 Classification Standards For Filtering at Least 95% of Airborne Particles
- Soft Inner Materials Enhance Comfort During Extended Wear
- Designed To Fit Well With Various Types of Eyewear
- Multiple Layers of Material To Enhance Filtration Efficiency
- Suitable For Use in Healthcare Settings, Including Surgical Applications
- Effective Filtration Protects Against Airborne Particles, Including Dust, Mists, and Fumes
- Exposure Control Helps Reduce the Risk of Contact With Infectious Agents
- Comfortable Wear Allows for Extended Use
Product Specifications
- Manufacturer: 3M
- Product Numbers: 1860, 1860S
- Sizes: Small, Regular
- Color: Teal
- Latex Indicator: Not Made With Natural Rubber Latex
- Application: Surgical Respirator
- Bacterial Filtration Efficiency (BFE): 99%
- Exhalation Valve: No
- Filter Class: KN95, N95, P2
- Flame-Resistant: No
- Fluid-Resistant: 120 mmHg
- Potential Hazards: Mold and Silica
- Nosefoam: Yes
- Shape: Cup
- Strap Type: Braided Comfort Strap
- Shelf Life: 5 Years
- Application: Particulate Respirator
- Industry: Healthcare
- Registered Medical Device: Yes
- eClass 14 Classification Group: 40220112
- Countries of Origin: USA and Singapore
Frequently Asked Questions (FAQs)
How do I ensure I’m wearing it properly?
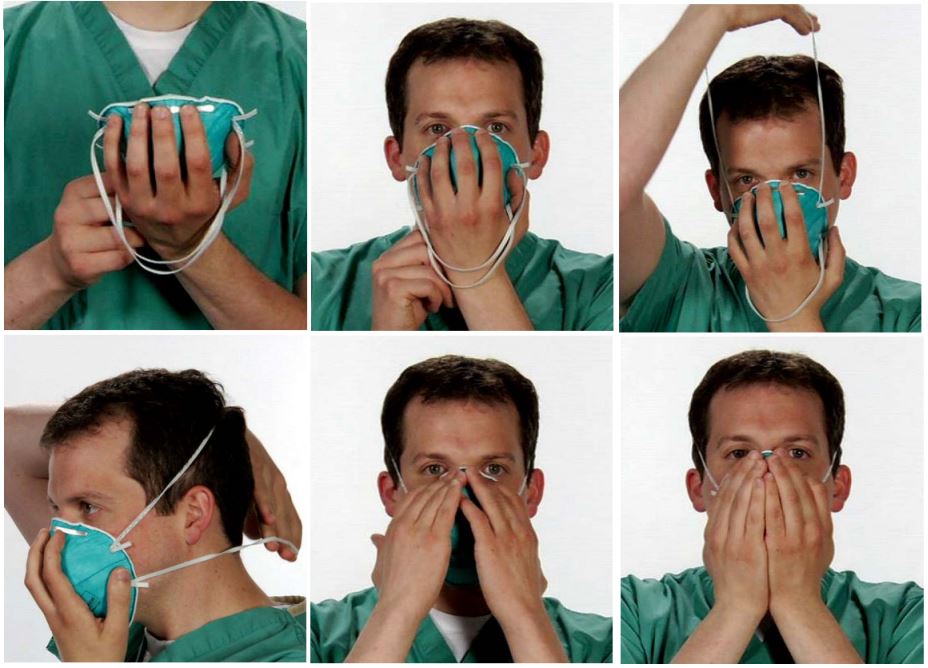
The mask should fit securely on the wearer’s face. To ensure optimal efficiency, follow the following steps when applying.
- With the mask resting in the palm of the hand, raise the mask up to the user’s face. The metal nose piece should line up with the bridge of the wearer’s nose and the mask should extend beneath the wearer’s chin.
- Pull the elastic bands over the wearer’s head. One elastic should rest above the user’s ears and the other below them.
- Bend the metal nose piece with both hands until it fits the shape of the wearer’s nose without leaving any gaps. It is important to use two hands for this step as only using one could result in unwanted gaps which could lessen the mask’s effectiveness.
- Test to ensure the mask is secure and there aren't any gaps between the mask and the wearer's face.
What is the shelf life?
These respirators can last up to 5 years in their unopened packaging if stored in temperatures between -4 and 86 degrees Fahrenheit with 80% relative humidity.
What is the difference between a face mask and a respirator?
Although both are used in medical professions, there are a few key differences between masks and respirators. Surgical masks are fluid-resistant and are intended to protect wearers from potentially harmful microorganisms transmitted from person to person as well as bodily fluids. In addition to fluid protection, respirators filter dangerous particles found in the air. They also have a more snug fit on the wearer’s face for added protection.
What is the Bacterial Filteration Efficiency (BFE)
This mask successfully filters 99% of airborne bacteria and protects the wearer’s health with maximum efficiency.
Additional Information
| Manufacturer | 3M |
|---|
Product Comparison

3M 1860 N95 Surgical Respirator Mask
$32.29
| 
Harley L-188 N95 Respirator Face Mask - NIOSH Approved
$2.15
| 
Moldex N95 Particulate Respirator Surgical Mask
$322.05
| 
PMT KN95 4.5 cm Thick Disposable Cone Mask
$1.50
| |
|---|---|---|---|---|
| Bacterial Filtration Efficiency (BFE) | 99.9% | 95% | 99.9% | 95% |
| Foldable | No | Yes | No | No |
| Sizes | Small, Regular | One Size | X-Small, Small, Medium, Large | One Size |
| Nose Piece | Flexible Metal Nose Ridge | Flexible Metal Nose Ridge | Sealable Nose Ridge | Flexible Metal Nose Ridge |
| Reviews | ||||
| View Details | View Details | View Details | View Details |












Login and Registration Form